
Cathode rays are normally invisible beams of particles that can be. Thomson was experimenting with cathode rays. 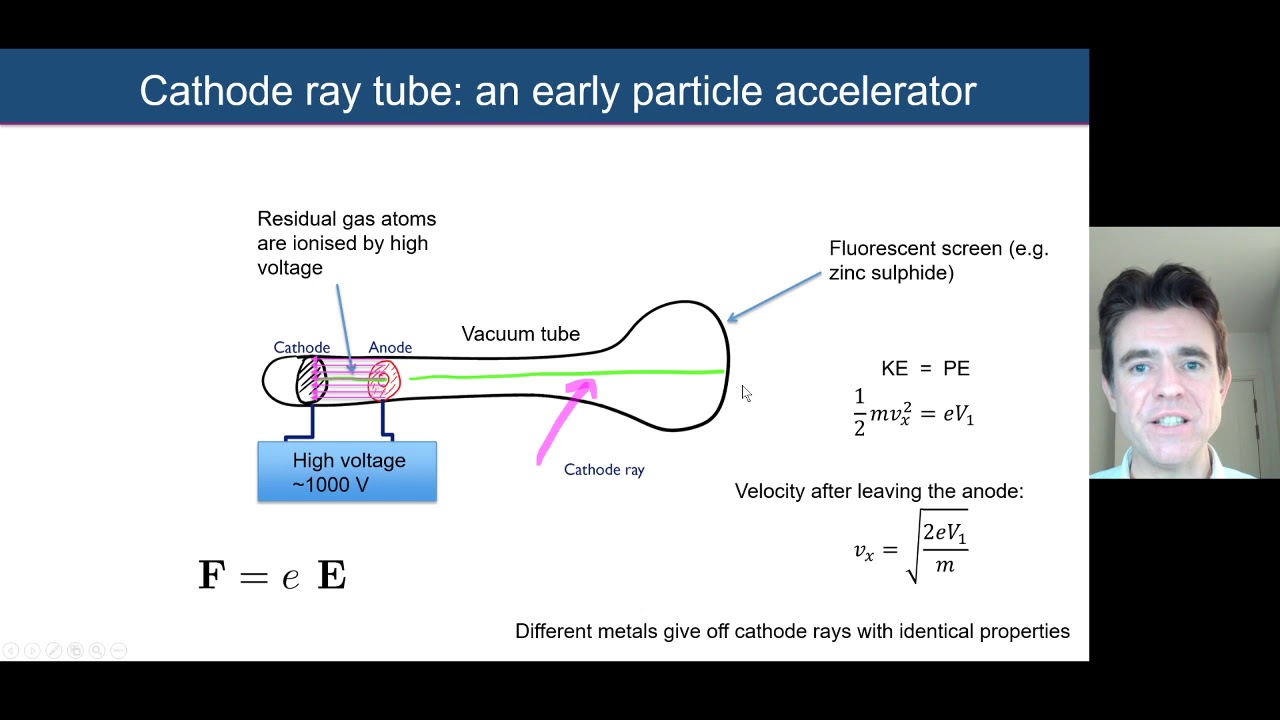
THE SMALLEST AND LIGHTEST POSITIVE ION WAS OBTAINED FROM HYDROGEN AND WAS CALLED PROTON. What Was the Cathode Ray Experiment Picture it: England, 1897. The behavior of these particles in a magnetic or electric field is opposite to that of electrons or cathode rays. Some positively charged particles carry a multiple of a fundamental unit of electric charge.Ĥ. The metal had two small diversions (slits), leading to an electrometer that could measure a small. He built his cathode ray tube with a metal cylinder on the other end. The charge to mass ratio of particles depends on the gas from which it originates.ģ. Thompson, conducted his first cathode ray tube experiment to prove that rays emitted from an electron gun are inseparable from the latent charge. Those are simply the positively charged gaseous ions.Ģ. 
The positively charged particles depend upon the nature of gas present in the cathode ray tube. The experiment for canal rays was carried out in modified cathode ray tube, by E. the cathode rays consist of electrons, while the anode/canal rays are the positively charged gaseous ions. You are right, both kinds of rays are emitted simultaneously. Thomson, Cathode rays (Friday evening meeting of the. The gold foil was surrounded by a detector screen that would flash when hit with an α \alpha α alpha particle. his corpuscle hypothesis, and in section 4 I discuss the experiments of 1897. Most of the radiation was absorbed by the lead, but a thin beam of α \alpha α alpha particles escaped out of the pinhole in the direction of the gold foil. Thompson, conducted his first cathode ray tube experiment to prove that rays emitted from an electron gun are inseparable from the latent charge. In this case, Rutherford placed a sample of radium (a radioactive metal) inside a lead box with a small pinhole in it. He then used a cathode ray tube to measure the. Alpha particles are helium nuclei ( 2 4 He 2 ) (_2^4\text) ( 2 4 He 2 ) left parenthesis, start subscript, 2, end subscript, start superscript, 4, end superscript, start text, H, e, end text, start superscript, 2, plus, end superscript, right parenthesis, and they are given off in various radioactive decay processes. By performing experiments with charged particles in gases, Thomson was able to estimate the magnitude of qe/me. In his famous gold foil experiment, Rutherford fired a thin beam of α \alpha α alpha particles (pronounced alpha particles) at a very thin sheet of pure gold. Narrow beam of cathode rays passing between two charged metal plates, striking a fluorescent screen. Charge to mass ratio(e/m) of an electron was determined by Thomson. Match J.J Thompsons Cathode Ray Experiment Description. The next groundbreaking experiment in the history of the atom was performed by Ernest Rutherford, a physicist from New Zealand who spent most of his career in England and Canada. Electrons were discovered using cathode ray discharge tube experiment.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
